This summary of my understanding of LOP operations. It is all based on John Deakin's Pelican's Perch column on AVweb. His LOP know-how is so spread around in those columns that I find it difficult put it all together, so hence this attempt to summarize. He now works at GAMI, and I am grateful to him for all the knowledge and graphs. I will try to put quotes around his words and not mine.
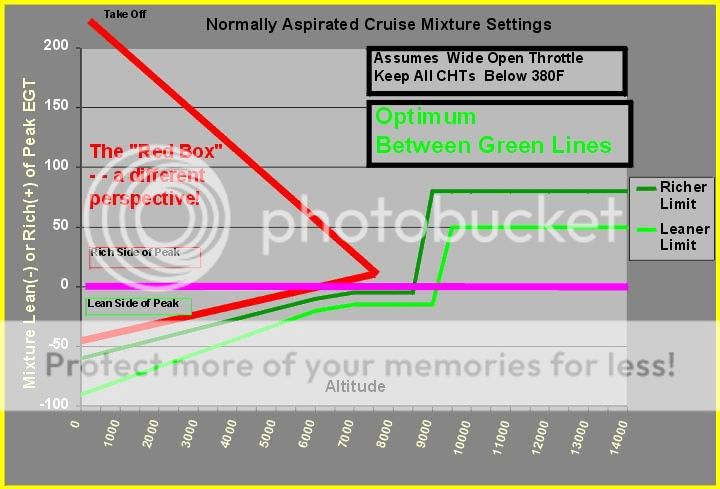
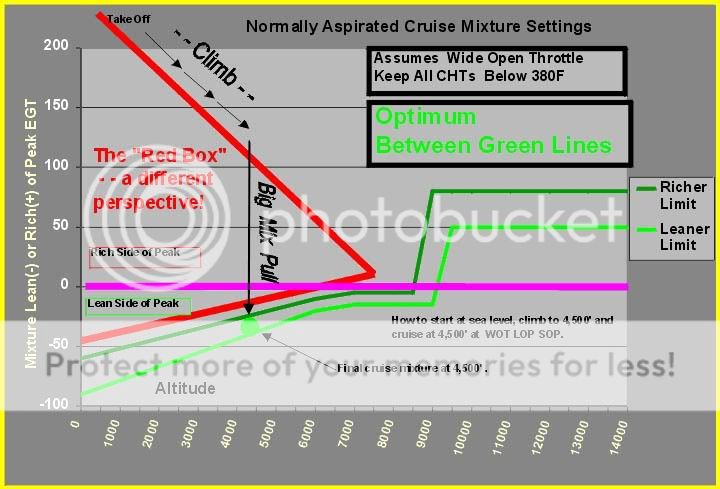
I am big advocate of LOP operations, and even do it at low altitude, like 700 ft MSL, and I hope this summary will clearly illustrate why this is safe and desirable.
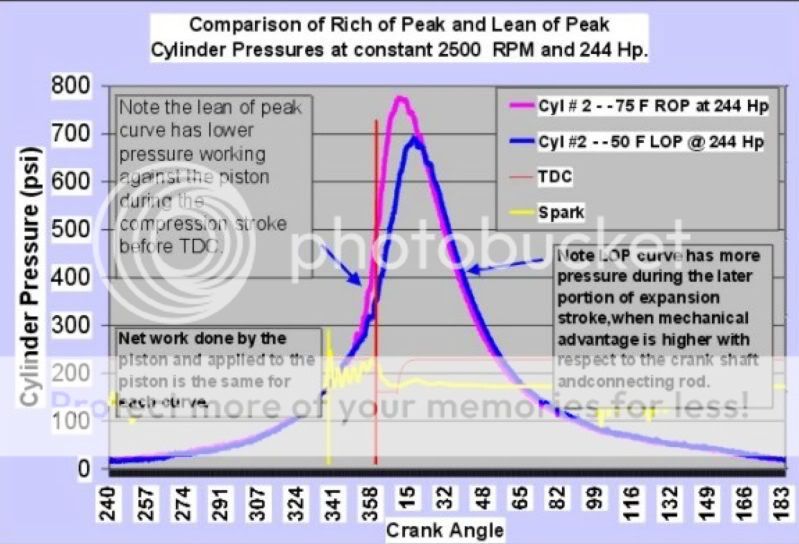
Let's start with a graph demonstrating the relationship between mixture, EGT, CHT, ICP and HP:

You can see that the Intra Cylinder Pressure tracks very well with the Cylinder Head Temperature. There is also a good correlation between those lines and the Exhaust Gas Temperature.
Key point from Pelican's Perch #65:
"On the rich side of peak, leaner is hotter, but on the lean side, leaner is cooler." That's a crucial concept! Repeat that to yourself, until you "get it.""
Temperature and Detonation:
Pelican's Perch #43:
"We know that combustion temperatures are in the 3,000ºF to 4,000ºF range, EGT "only" run around 1,600ºF, and CHTs down around 400ºF. How can this be? 4,000ºF is more than enough to melt steel, so how does the interior lining of the cylinder survive? Why don't we see hotter temperatures on our instruments? Why doesn't the aluminum piston melt down, when aluminum melts at less than 1,000ºF?
There is a thermal boundary layer, on the order of a millimeter thin or so, that acts as a buffer to protect the steel cylinder walls and the surface of the aluminum piston. Think of it as the thermal equivalent of the aerodynamic boundary layer out on your wing. The metal and the molecules right next to it will be at roughly the CHT reading or a bit higher, the next layers will be hotter and hotter, until the layer next to the combustion event will be at the combustion temperatures. That very thin thermal boundary layer acts as a nice insulation barrier, limiting the rate at which heat can be transferred from the bulk combustion gases into the interior walls of the cylinder head, cylinder barrel, and piston.
The heat transfer is continuous, as the heat moves first through the boundary layer, and then the cylinder wall and is finally carried away by the cooling airflow around the fins on the cylinders. Each intake stroke brings in a cool new charge, which starts the process all over again. There is also a matter of time of exposure. The high-pressure part of the combustion event takes up only about 40 degrees or so of crankshaft rotation, and the very hottest part of that only about 20 degrees, so during the other 700 degrees of crank rotation, cooler temperatures prevail. EGT shows only a number that represents a momentary flash of heat during a small portion of the combustion cycle (when the exhaust valve opens and exhaust gas flows across the EGT probe), and a rapidly dropping temperature at that.
This is NOT the major factor that determines how hot their exhaust valve is during operation. The events that happen a few degrees of crankshaft rotation earlier are much more significant because the temperatures are MUCH hotter than the piddling little 'ol 1500ºF measured by the EGT probe."
"We have nice cool induction air and fuel entering a cylinder;
The cylinder happens to have very hot walls. Those hot walls cause some of that nice cool induction air to start to heat up. And it doesn't all happen uniformly.
Further, shortly after the sparks go off, we have a couple of flame fronts, giving off lots of infrared heat, adding to the continuing heat load being absorbed by some of those little remote pockets of fuel and air that are waiting for the flame front to arrive and consume them;
The unburned mixture is experiencing a very rapid increase in pressure, because of two things: A) The piston is rising rapidly during the compression stroke; and B) the flame front combustion products are creating a huge increase in released energy and resulting bulk gas pressure, all of which is neatly measured on the pressure traces you see in the accompanying graphics.
At least some of those little "local pockets" of unburned combustion mixtures have exactly the right mixture of fuel and air to be just a hair-trigger away from exploding.
And … if the fuel is the wrong octane, or the spark advance was set too soon, or the manifold pressure was too high, or the cylinder head temperature was too high ... then one or more of those little "local pockets" of unburned fuel do just that ... they "explode."
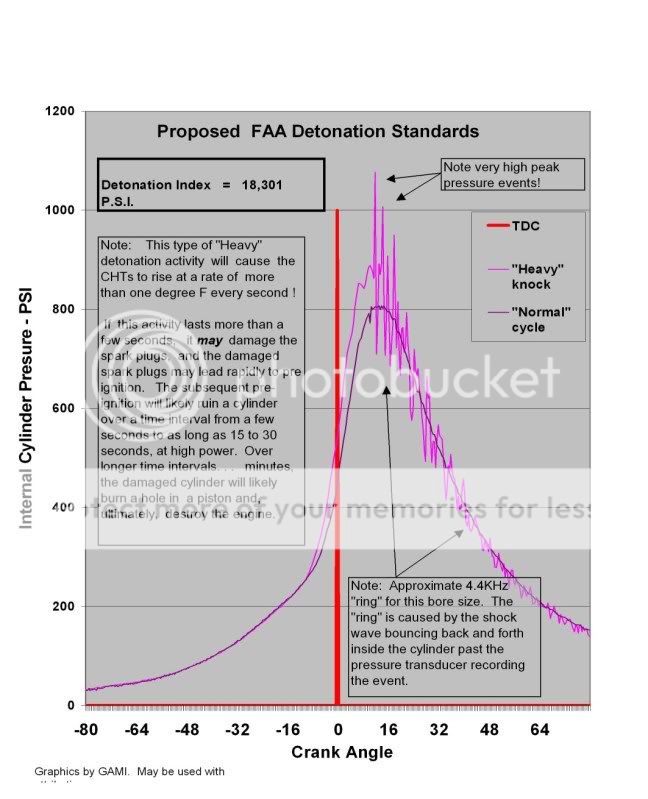
That is what we call "detonation".
Each explosion creates a shock wave that travels at the speed of sound (remember, the speed of sound inside the cylinder, at somewhere near 4000 degrees, is very much faster than at a standard day!) and bounces off the walls of the combustion chamber every 1/5th of a millisecond or so (giving off a 5KHz "ping" that you will not hear in the cockpit). Each of those explosions creates a very sharp rise in pressure and sets off a shock wave, which vibrates back and forth across the cylinder. This shock wave can be just the right amount of additional pressure to cause some other little remote local pocket of fuel and air to, in turn, explode, adding to the problem.
As detonation grows more serious, it will become audible, and this is the pinging you'll hear from that old auto engine on the uphill grade. Remember, you will NOT hear it on an aircraft engine."
Mild Detonation:
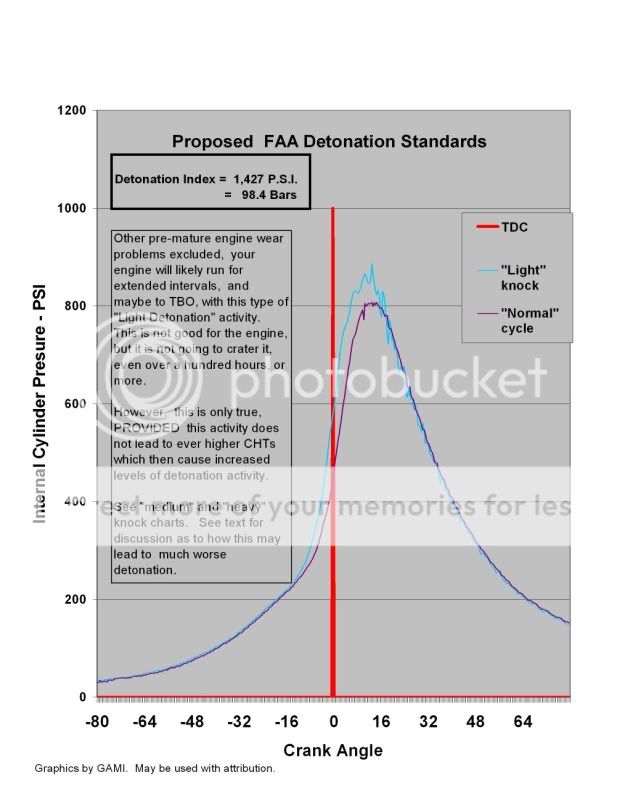
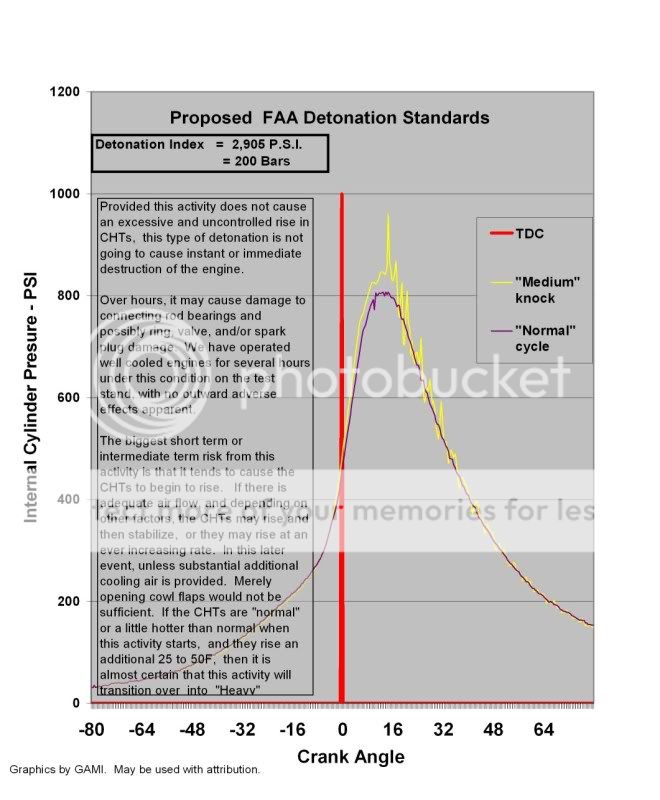
Moderate Detonation:
I am big advocate of LOP operations, and even do it at low altitude, like 700 ft MSL, and I hope this summary will clearly illustrate why this is safe and desirable.
Let's start with a graph demonstrating the relationship between mixture, EGT, CHT, ICP and HP:

You can see that the Intra Cylinder Pressure tracks very well with the Cylinder Head Temperature. There is also a good correlation between those lines and the Exhaust Gas Temperature.
Key point from Pelican's Perch #65:
"On the rich side of peak, leaner is hotter, but on the lean side, leaner is cooler." That's a crucial concept! Repeat that to yourself, until you "get it.""
Temperature and Detonation:
Pelican's Perch #43:
"We know that combustion temperatures are in the 3,000ºF to 4,000ºF range, EGT "only" run around 1,600ºF, and CHTs down around 400ºF. How can this be? 4,000ºF is more than enough to melt steel, so how does the interior lining of the cylinder survive? Why don't we see hotter temperatures on our instruments? Why doesn't the aluminum piston melt down, when aluminum melts at less than 1,000ºF?
There is a thermal boundary layer, on the order of a millimeter thin or so, that acts as a buffer to protect the steel cylinder walls and the surface of the aluminum piston. Think of it as the thermal equivalent of the aerodynamic boundary layer out on your wing. The metal and the molecules right next to it will be at roughly the CHT reading or a bit higher, the next layers will be hotter and hotter, until the layer next to the combustion event will be at the combustion temperatures. That very thin thermal boundary layer acts as a nice insulation barrier, limiting the rate at which heat can be transferred from the bulk combustion gases into the interior walls of the cylinder head, cylinder barrel, and piston.
The heat transfer is continuous, as the heat moves first through the boundary layer, and then the cylinder wall and is finally carried away by the cooling airflow around the fins on the cylinders. Each intake stroke brings in a cool new charge, which starts the process all over again. There is also a matter of time of exposure. The high-pressure part of the combustion event takes up only about 40 degrees or so of crankshaft rotation, and the very hottest part of that only about 20 degrees, so during the other 700 degrees of crank rotation, cooler temperatures prevail. EGT shows only a number that represents a momentary flash of heat during a small portion of the combustion cycle (when the exhaust valve opens and exhaust gas flows across the EGT probe), and a rapidly dropping temperature at that.
This is NOT the major factor that determines how hot their exhaust valve is during operation. The events that happen a few degrees of crankshaft rotation earlier are much more significant because the temperatures are MUCH hotter than the piddling little 'ol 1500ºF measured by the EGT probe."
"We have nice cool induction air and fuel entering a cylinder;
The cylinder happens to have very hot walls. Those hot walls cause some of that nice cool induction air to start to heat up. And it doesn't all happen uniformly.
Further, shortly after the sparks go off, we have a couple of flame fronts, giving off lots of infrared heat, adding to the continuing heat load being absorbed by some of those little remote pockets of fuel and air that are waiting for the flame front to arrive and consume them;
The unburned mixture is experiencing a very rapid increase in pressure, because of two things: A) The piston is rising rapidly during the compression stroke; and B) the flame front combustion products are creating a huge increase in released energy and resulting bulk gas pressure, all of which is neatly measured on the pressure traces you see in the accompanying graphics.
At least some of those little "local pockets" of unburned combustion mixtures have exactly the right mixture of fuel and air to be just a hair-trigger away from exploding.
And … if the fuel is the wrong octane, or the spark advance was set too soon, or the manifold pressure was too high, or the cylinder head temperature was too high ... then one or more of those little "local pockets" of unburned fuel do just that ... they "explode."
That is what we call "detonation".
Each explosion creates a shock wave that travels at the speed of sound (remember, the speed of sound inside the cylinder, at somewhere near 4000 degrees, is very much faster than at a standard day!) and bounces off the walls of the combustion chamber every 1/5th of a millisecond or so (giving off a 5KHz "ping" that you will not hear in the cockpit). Each of those explosions creates a very sharp rise in pressure and sets off a shock wave, which vibrates back and forth across the cylinder. This shock wave can be just the right amount of additional pressure to cause some other little remote local pocket of fuel and air to, in turn, explode, adding to the problem.
As detonation grows more serious, it will become audible, and this is the pinging you'll hear from that old auto engine on the uphill grade. Remember, you will NOT hear it on an aircraft engine."
Mild Detonation:

Moderate Detonation:

Last edited: